Our
Vision
To become a pre-eminent biological research & commercial institution committed to changing and saving lives.
Our
Mission
To provide the capacity for advanced research, innovation, and dissemination of new knowledge of commercial importance, while sustaining a mutually productive engagement with the local community.
Our Services
- We provide comprehensive R&D, preclinical & toxicity testing services to help clients from a wide range of disciplines.
- We provide high quality services to the industries and academia and private sector clients.
- We are extending our services throughout the Middle East and Asian countries.
- Our experts can customise study plans to meet numerous requirements for specialty projects meeting customer satisfaction.
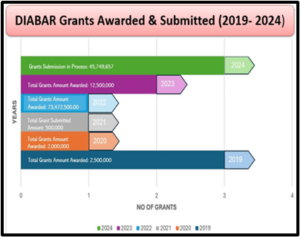
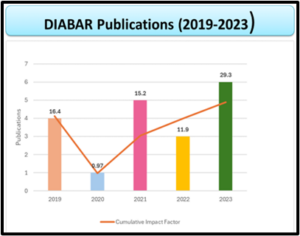
DIABAR: A Promise of Quality Excellence
Focused by our commitment in providing reliability, innovation and excellence in scientific discovery while safeguarding health & well-being of both humans and animals. Providing high-quality value-centred products and pioneering in Pre-Clinical & Toxicological Contract Laboratory testing services nation-wide. Learn more about our Quality Management System here.
Working with DIABAR
DIABAR is dedicated to developing innovative new products that deliver cutting-edge treatments through advanced therapeutics. These products are designed to enhance prevention, diagnosis, and treatment, providing our customers with access to comprehensive and integrated healthcare options.
Our studies include:
- Development of the indigenous Rabies vaccine formulation
- Production of indigenous Anti-Snake Venom (Hyperimmunized Plasma) as ‘Drug Substance’
- Preclinical Testing of Covid-19 Vaccine.
- Development and Validation of bio-/analytical test methods.
- Conducting stability studies in line with ICH guidelines.
- Formulating creams and ointments and wound patches.
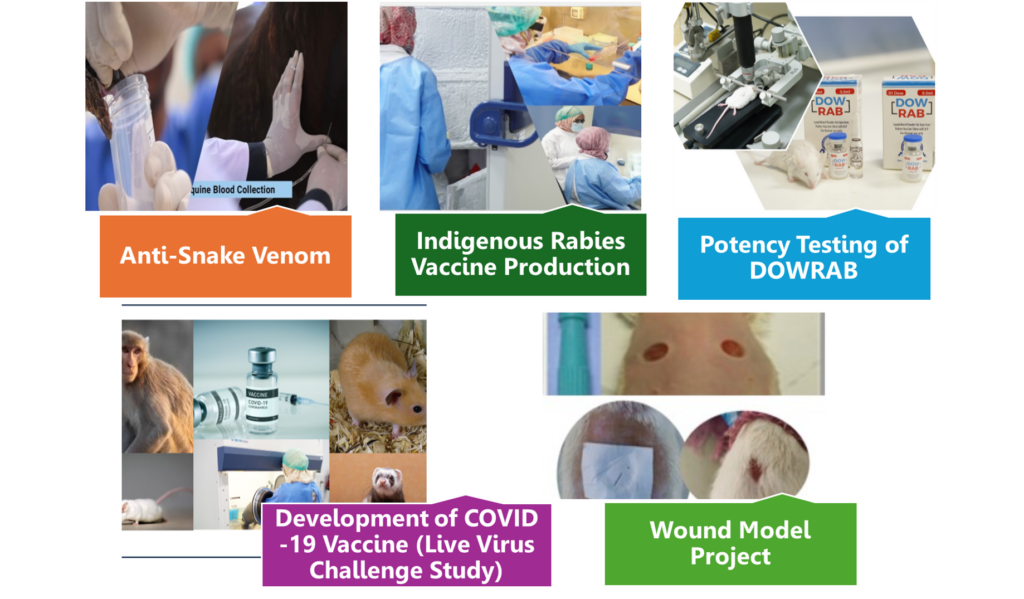
DIABAR is dedicated to upholding high standards across its manufacturing processes. The production of sophisticated, life-saving therapies using advanced technologies reflects both innovation and a commitment to health. The role of DIABAR’s Quality and Production teams is essential in ensuring that these products are safe and effective when they reach patients. DIABAR maintains a strong focus on quality and availability within the healthcare industry.
DIABAR-DUHS is actively involved in the production of Hyper-Immunized Plasma-ASV in accordance with international guidelines and the DIABAR-QMS control system. We have successfully completed four consecutive trial batches and one commercial batch of Hyper-Immunized Plasma-ASV. DIABAR staff are well-trained in the maintenance of hyper-immunized equines, the collection of plasma under aseptic conditions, and the processing, labeling, and storage of the final product. Furthermore, the establishment of the Basic Biological Manufacturing Facility – BSL III will support the bulk production and commercialization of vaccines, driving the self-sustaining production of biologicals and vaccine preparations in Pakistan, reducing reliance on international sources.
Currently, DIABAR is in the process of manufacturing anti-sera and vaccines, such as anti-rabies and anti-tetanus, at the R&D level. These efforts will pave the way for mass-scale production and technology transfer advancements.

- We transform new solutions to meet changing healthcare needs and create new methods that support the delivery of healthcare.
- Innovating solutions for healthcare not only addresses current challenges but also paves the way for a more efficient and impartial system.
- We are committed to protect the health and well-being of our employees to foster a positive work environment and enhance productivity. It can lead to higher job satisfaction and retention, ultimately benefiting the entire organisation.
- We earn trust of our clients through the highest standards of quality and safety, ethics and integrity.
- Together, we aim to inspire preventive action, promote essential health education, and create a lasting impact.
- We support Sustainable Development Goals (SDGs) 3, 4, and 17 by prioritising good health and well-being, quality education, and meaningful partnerships for sustainable development.
Our Management Team
- Rajab Ali – Phlebotomist
- Sarfaraz-ud-din Ahmed – Phlebotomist
- Gul Sher Umrani – Laboratory Technician
- Abdul Jabbar – Veterinary Assistant
- Ghulam Asghar – Veterinary Assistant
- Ghulam Hussain – Animal Attendant
- Fayaz Ali – Stable Boy
- Malhar-ud-din – Stable Boy
- Ali Raza – Stable Boy
- Basheer Ahmed – Stable Boy
- Basharat Ali – Animal Attendant
- Pervaiz Ali – Attendant
- Soofdal – Snake Handler
- Muhammad Bux – Stable Boy
- Jaral – Stable Boy
- Jamal Uddin – Attendant
- Shafiq Ahmed – Sweeper
- Imtiaz Ali – Animal Attendant
- Farrukh Rasheed – Animal Attendant
- Irfan Ali – Animal Attendant
- Qurban Ali – Animal Attendant
- Waqar Ali – Animal Attendant
- Naib Ali – Animal Attendant
- Lal Dino – Animal Attendant
- Jansar – Animal Attendant
Our Collaboration / Clients
















Picture Gallery
You can view our Picture Gallery here.
Latest News
Contact Us
DOW INSTITUTE FOR ADVANCED BIOLOGICAL & ANIMAL RESEARCH (DIABAR)